Application of 3D printing technology-assisted individualized combined stent in type A aortic dissection surgery
-
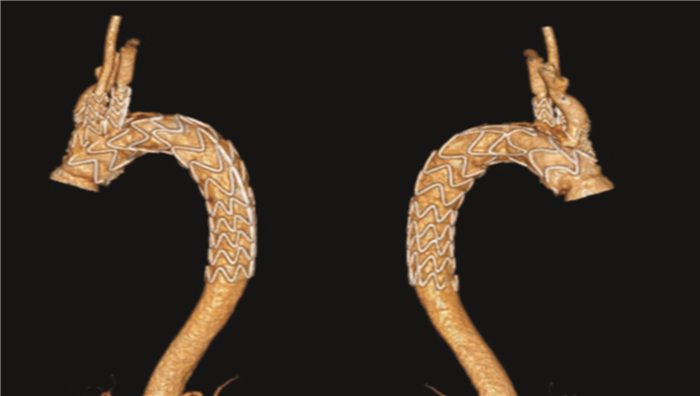
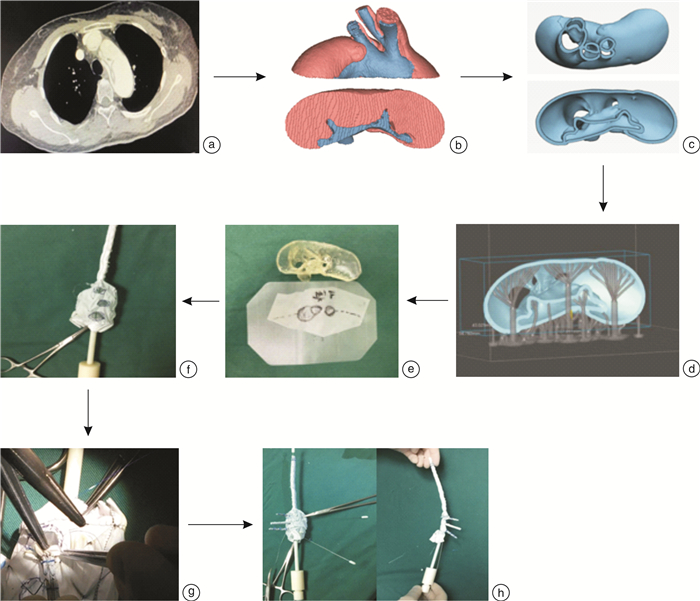
摘要: 目的 探讨3D打印技术辅助的个体化组合支架在Stanford A型主动脉夹层外科治疗中的效果,并与传统孙氏手术进行对比分析。方法 选取2017年9月—2021年7月安徽医科大学第二附属医院收治住院的37例Stanford A型主动脉夹层患者资料,按手术方式分为3D打印技术辅助的个体化支架手术组(观察组)17例和孙氏手术组(对照组)20例。术前患者均进行心脏超声、主动脉CT血管造影(CTA)、肝肾功能、血电解质、血常规等检查。统计并分析患者手术及体外循环相关数据,术后统计住院时间、ICU监护时间等指标。患者术后1个月进行临床随访,复查超声心动图及主动脉CTA。结果 2组患者手术顺利,术中无死亡。对照组和观察组手术时间[(377.70±80.35) min vs(311.18±74.53) min,P < 0.05]、体外循环时间[(168.80±24.28) min vs(147.71±24.77) min,P < 0.05]、术中停循环时间[(18.70±4.04) min vs(9.12±4.90) min,P < 0.05]、ICU监护治疗时间[(7.61±1.79) d vs(6.25±1.29) d,P < 0.05]、呼吸机使用时间[(4.46±1.52) d vs(3.03±0.77) d,P < 0.05]、术后住院时间[(19.06±5.71) d vs(15.69±1.96) d,P < 0.05]比较,差异均有统计学意义。观察组与对照组均全部一次关胸,无二次手术。术后对照组死亡2例,治愈出院18例,观察组自动出院1例,治愈出院16例。出院患者术后1个月随访无死亡病例,所有患者均复查主动脉CTA及心脏超声,结果提示主动脉弓、降主动脉血流通畅,支架血管位置正常,扩张良好,无位移、狭窄,支架周围无造影剂外渗,心功能良好。结论 3D打印技术辅助的个体化组合支架治疗Stanford A型主动脉夹层效果较好,可减少手术时间、体外循环时间及术中停循环时间。在降低手术难度的同时,减少了术后并发症的发生,有助于降低患者死亡率,术后早期复查效果满意。
-
关键词:
- 主动脉夹层 /
- Stanford A型 /
- 个体化组合支架 /
- 3D打印 /
- 停循环时间
Abstract: Objective To discuss the effect of 3D printing technology-assisted individualized combined stent in the surgical treatment of Stanford A-type aortic dissection, and compare it with traditional Sun's surgery.Methods The data of 37 patients with Stanford type A aortic dissection admitted to the hospital from September 2017 to July 2021 were selected. According to the operation method, they were divided into 3D printing technology-assisted individualized combined stent surgery group (observation group) with 17 cases and Sun's surgery group (control group) with 20 cases. Before surgery, all patients underwent cardiac ultrasound, aortic angiography, liver and kidney function, blood electrolyte, blood routine, and other inspections and tests. Observation data related to patients' surgery and cardiopulmonary bypass, as well as postoperative hospitalization time, ICU monitoring time, and other observation indicators were counted and analyzed. The patient underwent clinical follow-up 1 month after surgery and rechecked echocardiography and aortic CTA.Results The operation of patients in two groups was successful, and there was no death during the operation. In the control group and observation group, operation time[(377.70±80.35) min vs(311.18±74.53) min, P < 0.05], cardiopulmonary bypass time[(168.80±24.28) min vs(147.71±24.77) min, P < 0.05], intraoperative circulatory arrest time[(18.70±4.04) min vs(9.12±4.90) min, P < 0.05], ICU monitoring and treatment time[(7.61±1.79) d vs(6.25±1.29) d, P < 0.05], respiration machine usage time[(4.46±1.52) d vs(3.03±0.77) d, P < 0.05], postoperative hospital stay[(19.06±5.71) d vs(15.69±1.96) d, P < 0.05]were different significantly. Both the observation group and the control group had their chests closed at one time without secondary surgery. After the operation, 2 cases died and18 cases were cured and discharged in the control group. One case was discharged automatically and 16 cases were cured and discharged in the observation group. The discharged patients were followed up for 1 month after the operation, and there was no death. All patients were rechecked with aortic CTA and echocardiography. The results suggested that the blood flow of the aortic arch and descending aorta were smooth. The position of the stent was normal. The expansion was good. There was no displacement and stenosis. There was no extravasation of contrast agent around the stent, and the heart function was good.Conclusion The personalized combined stent assisted by 3D printing technology is effective in treating Stanford A-type aortic dissection, and the operation time, extracorporeal circulation time, and intraoperative circulatory arrest time are short. While reducing the difficulty of surgery, it also reduces the probability of postoperative complications. The results of early postoperative reexamination are satisfactory. -

-
表 1 观察组与对照组术中情况比较
Table 1. Comparison of intraoperative conditions between the observation group and the control group
例(%), X±S 项目 对照组(20例) 观察组(17例) P 手术时间/min 377.70±80.35 311.18±74.53 0.014 体外循环时间/min 168.80±24.28 147.71±24.77 0.013 术中停循环时间/min 18.70±4.04 9.12±4.90 < 0.001 主动脉瓣置换 13(65.00) 11(64.71) 0.627 表 2 观察组与对照组患者术后一般情况及并发症情况比较
Table 2. Comparison of postoperative general condition and complications between the observation group and the control group
例(%), X±S 项目 对照组(20例) 观察组(17例) P 术后ICU监护时间/d 7.61±1.79 6.25±1.29 0.017 呼吸机使用时间/d 4.46±1.52 3.03±0.77 0.002 术后住院时间/d 19.06±5.71 15.69±1.96 0.032 肺部感染 4(20.00) 3(17.65) 0.596 急性肾衰竭 4(20.00) 2(11.76) 0.413 苏醒延迟 2(10.00) 1(5.88) 0.562 急性呼吸衰竭 1(5.00) 0(0) 0.541 截瘫 0(0) 0(0) 二次开胸 0(0) 0(0) 死亡或自动出院 2(10.00) 1(5.88) 0.562 注:肺部感染:血象持续升高,痰细菌培养呈阳性;急性肾衰竭:术后因少尿、血肌酐升高和高钾血症等原因使用肾脏替代治疗;苏醒延迟:患者苏醒时间术后超过12 h,且Ramsay评分≥5;急性呼吸衰竭:出现呼吸困难、呼吸急促等表现,且动脉血氧分压 < 60 mmHg(1 mmHg=0.133 kPa)。 -
[1] Wu Y, Jiang R, Xu P, et al. Perioperative Results and Risk Factors for In-Hospital Mortality In Patients With Stanford Type A Aortic Dissection Undergoing Sun's Procedure-A Single Center Study[J]. Heart Surg Forum, 2018, 21(6): E432-E437. doi: 10.1532/hsf.1909
[2] Pitt MP, Bonser RS. The natural history of thoracic aortic aneurysm disease: an overview[J]. J Card Surg, 1997, 12(2 Suppl): 270-278.
[3] 孙立忠, 李建荣. 我国Stanford A型主动脉夹层诊疗进展与挑战[J]. 中华外科杂志, 2017, 55(4): 241-244. https://www.cnki.com.cn/Article/CJFDTOTAL-ZJYE201719004.htm
[4] Hagan PG, Nienaber CA, Isselbacher EM, et al. The International Registry of Acute Aortic Dissection(IRAD): new insights into an old disease[J]. JAMA, 2000, 283(7): 897-903. doi: 10.1001/jama.283.7.897
[5] Elsayed RS, Cohen RG, Fleischman F, et al. Acute Type A Aortic Dissection[J]. Cardiol Clin, 2017, 35(3): 331-345. doi: 10.1016/j.ccl.2017.03.004
[6] Luo J, Fu X, Zhou Y, et al. Aortic Remodeling Following Sun's Procedure for Acute Type A Aortic Dissection[J]. Med Sci Monit, 2017, 23: 2143-2150. doi: 10.12659/MSM.900345
[7] Ghazy T, Mahlmann A, Fajfrova Z, et al. Anastomotic leak after surgical repair of type A aortic dissection-prevalence and consequences in midterm follow-up[J]. Vasa, 2017, 46(5): 377-382. doi: 10.1024/0301-1526/a000644
[8] 焦瑞, 刘楠. 急性Stanford A型主动脉夹层术后急性肾损伤患者连续性肾脏替代治疗后的死亡预后因素分析[J]. 中华外科杂志, 2017, 55(4): 270-273. doi: 10.3760/cma.j.issn.0529-5815.2017.04.007
[9] 弓华, 张海燕, 靳津鸽. 急性Standford A型主动脉夹层围手术期患者发生低血氧症的危险因素及其预测价值[J]. 中国现代医学杂志, 2020, 30(2): 44-49. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXDY202002009.htm
[10] 喻雪飞, 董柱, 张本, 等. 急性Stanford A型主动脉夹层术后感染的危险因素分析及预防策略[J]. 临床心血管病杂志, 2021, 37(8): 758-763. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB202108017.htm
[11] 刘艳, 石晓卉, 肖东. 急性Stanford A型主动脉夹层患者主动脉弓部手术后脑部并发症发病情况及危险因素分析[J]. 中国现代医学杂志, 2016, 26(11): 98-101. https://www.cnki.com.cn/Article/CJFDTOTAL-TWXH202201002.htm
[12] 朱悦倩, 杨菁, 孙煦, 等. 急性Stanford A型夹层术后谵妄的危险因素分析[J]. 实用医学杂志, 2020, 36(7): 940-943. https://www.cnki.com.cn/Article/CJFDTOTAL-SYYZ202007025.htm
[13] Olsson C, Hillebrant CG, Liska J, et al. Mortality in acute type A aortic dissection: validation of the Penn classification[J]. Ann Thorac Surg, 2011, 92(4): 1376-1382. doi: 10.1016/j.athoracsur.2011.05.011
[14] Hadeed K, Acar P, Dulac Y, et al. Cardiac 3D printing for better understanding of congenital heart disease[J]. Arch Cardiovasc Dis, 2018, 111(1): 1-4.
-




 下载:
下载: