A retrospective controlled study of the effect of tirofiban-induced thrombocytopenia on the prognosis of patients
-
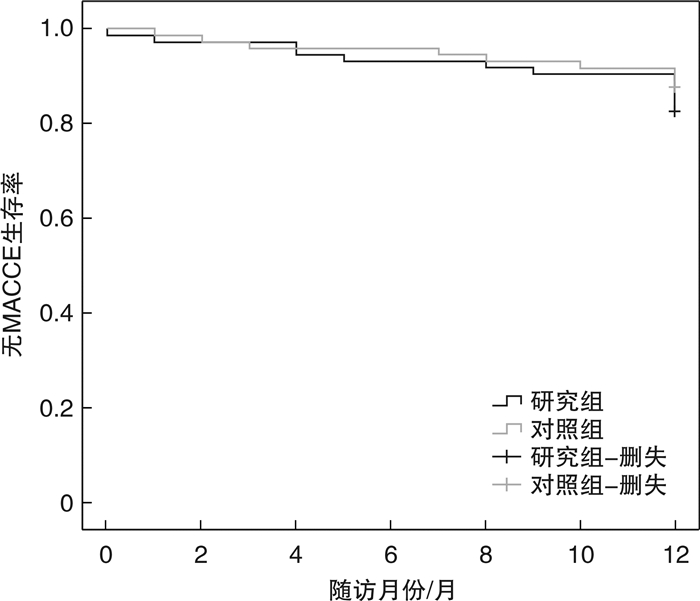
摘要: 目的 探讨经皮冠状动脉介入治疗(PCI)围手术期替罗非班诱导血小板减少症患者的预后情况。方法 选取郑州大学第一附属医院2016年1月1日—2019年12月30日收治入院的74例PCI围术期发生替罗非班诱导血小板减少的患者作为研究组;连续入选郑州大学第一附属医院2018年1月1日—2018年12月30日入院的213例PCI围术期使用替罗非班未发生血小板减少的患者作为对照组。采用倾向性评分匹配获得基线项目相似的对照组和研究组,对比2组30 d内出血事件及随访12个月时主要不良心脑血管事件(MACCE)。结果 匹配前研究组较对照组,年龄偏大[63(57,69)岁∶60(52,66)岁],有吸烟史的患者所占比例偏大[37(50.0)%∶64(30.0)%],血小板基数偏低[192(164,249)×109/L∶219(181,256)×109/L],且差异均有统计学意义(P< 0.05)。匹配后获得74例研究组和74例对照组,组间的基线资料比较差异无统计学意义(P>0.05)。与对照组比较,研究组30 d内总出血事件和轻微出血发生概率显著升高(P< 0.05)。随访12个月时研究组和对照组间的MACCE发生率比较差异无统计学意义(P>0.05)。结论 替罗非班诱导的血小板减少症增加30 d内出血的风险但不增加严重出血的风险,不增加随访中期MACCE发生的风险。Abstract: Objective To investigate the prognosis of patients with thrombocytopenia induced by tirofiban during the perioperative period of PCI.Methods The 74 patients who suffered from thrombocytopenia induced by tirofiban during the perioperative period of PCI were enrolled as a study group from January 1, 2016 to December 30, 2019 in the First Affiliated Hospital of Zhengzhou University. The 213 patients without thrombocytopenia who used tirofiban during the perioperative period of PCI were enrolled as a control group from January 1, 2018 to December 30, 2018 in the First Affiliated Hospital of Zhengzhou University. The control group and the study group with similar baseline items were selected by propensity score matching. Bleeding events within 30 days and major adverse cardiac and cerebrovascular events (MACCE) at 12 months of follow-up were compared between the two groups.Results Compared with the control group before matching, the age was older [63 (57, 69) years∶60 (52, 66) years], and the proportion of smoking history was higher [37 (50.0)%∶64 (30.0) %], the platelet base was low[192 (164, 249) × 109/L∶219 (181, 256) × 109/L] in the study group, and the difference was statistically significant (P< 0.05). After matching, 74 cases in the study group and 74 cases in the control group were obtained, and there was no significant difference in baseline data between the groups (P>0.05). Compared with the control group, the probability of total bleeding events and minor bleeding within 30 days in the study group was significantly increased (P< 0.05). There was no significant difference in the incidence of MACCE between the study group and the control group at 12 months of follow-up (P>0.05).Conclusion Tirofiban-induced thrombocytopenia increased the risk of bleeding within 30 days but did not increase the risk of severe bleeding. There was no increased risk of MACCE at mid-follow-up.
-
Key words:
- tirofiban /
- thrombocytopenia /
- bleeding /
- ischemia
-

-
表 1 研究组与对照组基线资料比较(PSM前)
Table 1. Comparison of baseline data between study group and control group(before PSM)
例(%), M(Q1, Q3) 项目 研究组(74例) 对照组(213例) P 年龄/岁 63(57,69) 60(52,66) 0.017 男性 54(73.0) 146(68.5) 0.475 体重/kg 70(60,78) 70(62,77) 0.416 既往史 高血压病 45(60.8) 122(57.3) 0.595 脑血管病 11(14.9) 35(16.4) 0.752 糖尿病 26(35.1) 54(25.4) 0.106 吸烟史 37(50.0) 64(30.0) 0.002 饮酒史 16(21.6) 34(16.0) 0.269 心肌梗死史 7(9.5) 22(10.3) 0.831 PCI史 11(14.9) 22(10.3) 0.292 冠状动脉移植史 1(1.4) 4(1.9) 1.000 KillipⅢ/Ⅳ级 17(23.0) 30(14.1) 0.075 左心室射血分数/% 61(56,64) 61(58,64) 0.614 收缩压/mmHg 130(116,144) 132(120,142) 0.312 舒张压/mmHg 77(69,86) 76(70,85) 0.555 白细胞数/(×109·L-1) 6.95(5.48,8.41) 6.80(5.70,8.20) 0.833 血小板基数/(×109·L-1) 192(164,249) 219(181,256) 0.024 肌酐/(μmol·L-1) 71.90(59.00,87.00) 70.60(60.80,83.00) 0.614 肾小球滤过率/(mL·min-1) 92.52(79.95,99.57) 94.42(82.20,102.62) 0.321 谷丙转氨酶/(U·L-1) 28.50(15.00,46.00) 24(16,37) 0.385 谷草转氨酶/(U·L-1) 26(17,49) 23(16,32) 0.053 总胆固醇/(mmol·L-1) 3.63(3.07,4.71) 3.81(3.37,4.59) 0.158 高密度脂蛋白/(mmol·L-1) 0.97(0.78,1.20) 1.00(0.86,1.20) 0.307 低密度脂蛋白/(mmol·L-1) 2.03(1.65,3.01) 2.34(1.88,3.01) 0.132 ACS分型 0.054 不稳定型心绞痛 45(60.8) 255(72.8) 急性心肌梗死 29(39.2) 58(27.2) 冠状动脉情况 左主干病变 8(10.8) 42(19.7) 0.082 单支病变 10(13.5) 29(13.6) 0.982 双支病变 16(21.6) 49(23.0) 0.807 3支病变 40(54.1) 93(43.7) 0.122 表 2 研究组与对照组基线项目对比(PSM后)
Table 2. Comparison of baseline data between study group and control group(after PSM)
例(%), M(Q1, Q3), X±S 项目 研究组(74例) 对照组(74例) P 年龄/岁 63(57,69) 61(55,68) 0.401 男性 54(73.0) 55(74.3) 0.852 体重/kg 70(60,78) 70(62,80) 0.432 既往史 高血压病 45(60.8) 45(60.8) 1.000 脑血管病 11(14.9) 11(14.9) 1.000 糖尿病 26(35.1) 17(23.0) 0.103 吸烟史 37(50.0) 39(52.7) 0.742 饮酒史 16(21.6) 19(257) 0.562 心肌梗死史 7(9.5) 8(10.8) 0.785 PCI史 11(14.9) 12(16.2) 0.821 冠状动脉移植史 1(1.4) 2(2.7) 1.000 KillipⅢ/Ⅳ级 17(23.0) 16(21.6) 0.843 左心室射血分数/% 61(56,64) 61(58,64) 0.988 收缩压/mmHg 130±20 129±18 0.862 舒张压/mmHg 77±12 76±13 0.989 白细胞数/(×109·L-1) 6.95(5.48,8.41) 6.80(5.60,8.16) 0.797 血小板基数/(×109·L-1) 192(164,249) 199(168,238) 0.723 肌酐/(μmol·L-1) 71.90(59.00,87.00) 69.90(62.75,82.45) 0.675 肾小球滤过率/(mL·min-1) 92.52(79.95,99.57) 94.72(82.11,102.64) 0.480 谷丙转氨酶/(U·L-1) 28.50(15.00,46.00) 24.60(17.75,35.50) 0.645 谷草转氨酶/(U·L-1) 26(17,49) 24(18,40) 0.535 总胆固醇/(mmol·L-1) 3.63(3.07,4.71) 3.76(3.25,4.26) 0.539 甘油三酯/(mmol·L-1) 1.30(0.93,1.69) 1.42(1.11,1.98) 0.164 高密度脂蛋白/(mmol·L-1) 0.97(0.78,1.20) 1.01(0.88,1.21) 0.246 低密度脂蛋白/(mmol·L-1) 2.03(1.65,3.01) 2.28(1.89,2.72) 0.274 ACS分型 0.052 不稳定型心绞痛 45(60.8) 56(75.7) 急性心肌梗死 29(39.2) 18(24.3) 冠状动脉情况 左主干病变 8(10.8) 10(13.5) 0.615 单支病变 10(13.5) 9(12.2) 0.806 双支病变 16(21.6) 15(20.3) 0.840 3支病变 40(54.1) 40(54.1) 1.000 表 3 30 d内出血事件比较
Table 3. Comparison of bleeding events within 30 days
例(%) 项目 研究组(74例) 对照组(74例) P 院内出血 30(40.5) 4(5.4) < 0.001 轻微出血 24(32.3) 4(5.4) < 0.001 小出血 5(6.8) 0(0) 0.069 大出血 1(1.4) 0(0) 1.000 表 4 随访12个月时两组MAACE比较
Table 4. Comparison of MAACE at 12 months follow-up
例(%) 组别 心绞痛复发住院 靶血管再次血运重建 支架内血栓形成 卒中 死亡 合计 研究组(74例) 4(5.4) 5(6.7) 1(1.4) 1(1.4) 2(2.7) 13(17.6) 对照组(74例) 1(1.3) 3(4.1) 1(1.4) 2(2.7) 2(2.7) 9(12.2) P 0.363 0.716 1.000 1.000 1.000 0.355 -
[1] Crea F, Libby P. Acute Coronary Syndromes: The Way Forward From Mechanisms to Precision Treatment[J]. Circulation, 2017, 136(12): 1155-1166. doi: 10.1161/CIRCULATIONAHA.117.029870
[2] 庞军, 吴强, 张钲, 等. 中国人群血小板抵抗与血小板糖蛋白受体基因相关性的荟萃分析[J]. 临床心血管病杂志, 2019, 35(1): 10-14. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB201901004.htm
[3] Friedland S, Eisenberg MJ, Shimony A. Meta-analysis of randomized controlled trials of intracoronary versus intravenous administration of glycoprotein Ⅱb/Ⅲa inhibitors during percutaneous coronary intervention for acute coronary syndrome[J]. Am J Cardiol, 2011, 108(9): 1244-1251. doi: 10.1016/j.amjcard.2011.06.039
[4] Valgimigli M, Campo G, de Cesare N, et al. Intensifying platelet inhibition with tirofiban in poor responders to aspirin, clopidogrel, or both agents undergoing elective coronary intervention: results from the double-blind, prospective, randomized Tailoring Treatment with Tirofiban in Patients Showing Resistance to Aspirin and/or Resistance to Clopidogrel study[J]. Circulation, 2009, 119(25): 3215-3222. doi: 10.1161/CIRCULATIONAHA.108.833236
[5] Li L, Ge Z, Zhang D, et al. Efficacy and safety of quadruple therapy including tirofiban in the treatment of Chinese NSTE-ACS patients failing to receive timely percutaneous coronary intervention[J]. Drug Des Devel Ther, 2017, 11: 3299-3307. doi: 10.2147/DDDT.S138714
[6] 唐茜, 王玉, 程景林. 替罗非班联合呋塞米对急性心肌梗死伴心力衰竭患者行急诊PCI后临床预后的影响[J]. 临床急诊杂志, 2021, 22(7): 441-444. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZLC202107001.htm
[7] Merlini PA, Rossi M, Menozzi A, et al. Thrombocytopenia caused by abciximab or tirofiban and its association with clinical outcome in patients undergoing coronary stenting[J]. Circulation, 2004, 109(18): 2203-2206. doi: 10.1161/01.CIR.0000127867.41621.85
[8] Bougie DW, Wilker PR, Wuitschick ED, et al. Acute thrombocytopenia after treatment with tirofiban or eptifibatide is associated with antibodies specific for ligand-occupied GPⅡb/Ⅲa[J]. Blood, 2002, 100(6): 2071-2076. doi: 10.1182/blood.V100.6.2071
[9] 马慧慧, 桑海强, 李云鹏, 等. 急性冠状动脉综合征患者PCI围术期肝素或替罗非班诱导血小板减少症的临床观察及干预分析[J]. 临床心血管病杂志, 2021, 37(4): 340-345. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB202104010.htm
[10] 中国医师协会心血管内科医师分会血栓防治专业委员会, 《中华医学杂志》编辑委员会. 肝素诱导的血小板减少症中国专家共识(2017)[J]. 中华医学杂志, 2018, 98(6): 408-417. doi: 10.3760/cma.j.issn.0376-2491.2018.06.003
[11] 郑丹. 3种方法对肝素诱导血小板减少症抗体的检测效能比较[J]. 临床血液学杂志, 2020, 33(10): 709-712. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202010012.htm
[12] Cuker A, Gimotty PA, Crowther MA, et al. Predictive value of the 4Ts scoring system for heparin-induced thrombocytopenia: a systematic review and meta-analysis[J]. Blood, 2012, 120(20): 4160-4167. doi: 10.1182/blood-2012-07-443051
[13] Mehran R, Rao SV, Bhatt DL, et al. Standardized bleeding definitions for cardiovascular clinical trials: a consensus report from the Bleeding Academic Research Consortium[J]. Circulation, 2011, 123(23): 2736-2747. doi: 10.1161/CIRCULATIONAHA.110.009449
[14] Yi YH, Yin WJ, Gu ZC, et al. A Simple Clinical Pre-procedure Risk Model for Predicting Thrombocytopenia Associated With Periprocedural Use of Tirofiban in Patients Undergoing Percutaneous Coronary Intervention[J]. Front Pharmacol, 2018, 9: 1456.
[15] Chen CY, Lee MY, Lin KD, et al. Diabetes mellitus increases severity of thrombocytopenia in dengue-infected patients[J]. Int J Mol Sci, 2015, 16(2): 3820-3830. doi: 10.3390/ijms16023820
[16] Mondal NK, Chen Z, Trivedi JR, et al. Oxidative stress induced modulation of platelet integrin á2ba3 expression and shedding may predict the risk of major bleeding in heart failure patients supported by continuous flow left ventricular assist devices[J]. Thromb Res, 2017, 158: 140-148. doi: 10.1016/j.thromres.2017.09.006
[17] Rubinfeld GD, Smilowitz NR, Berger JS, et al. Association of Thrombocytopenia, Revascularization, and In-Hospital Outcomes in Patients with Acute Myocardial Infarction[J]. Am J Med, 2019, 132(8): 942-948.e5. doi: 10.1016/j.amjmed.2019.04.003
[18] Salter BS, Weiner MM, Trinh MA, et al. Heparin-Induced Thrombocytopenia: A Comprehensive Clinical Review[J]. J Am Coll Cardiol, 2016, 67(21): 2519-2532.
-




 下载:
下载: