The role of cardiac resident macrophages in myocardial infarction and ischemia-reperfusion injury
-
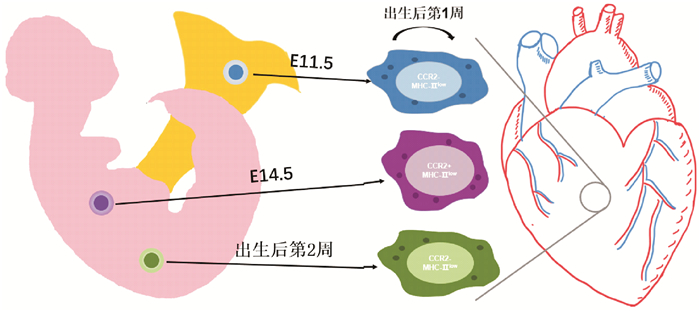
摘要: 传统观点认为,组织巨噬细胞由循环系统中骨髓来源的单核细胞分化产生,近期研究通过单细胞测序、遗传谱系示踪(genetic fate mapping and lineage tracing)等技术探究心脏巨噬细胞的分群及起源,发现心脏中的部分巨噬细胞在胚胎期即进入心脏,由卵黄囊中的前体细胞、胎肝单核细胞分化形成,并通过原位增殖维持细胞数目,该类细胞被称为心脏原位巨噬细胞。心脏原位巨噬细胞在稳态和损伤后心脏中发挥免疫作用,参与心肌缺血及恢复血流后的再灌注损伤过程中的炎症反应。本文结合最新研究进展,介绍了心脏原位巨噬细胞的来源及分型,并主要就心脏原位巨噬细胞在心肌梗死及血管再通导致的缺血再灌注损伤中发挥的作用作一综述,旨在为改善心肌梗死的临床预后提供新的思路。Abstract: The long-held notion was that tissue macrophages replenish from circulation monocytes. However, evidence has established that the majority of macrophages are established prenatally, originating from yolk sac progenitors and fetal liver monocytes, and maintained via in situ proliferation by using genetic fate mapping and lineage tracing techniques to investigate the origin of the cardiac macrophages. For their embryonic origin and self-renewal capacity, this population is defined as cardiac resident macrophage (CRM). CRMs are involved in the immune response in steady and following the disruption of homeostasis, including their specific role and contribution in the process of myocardial infarction and ischemia-reperfusion injury. According to the latest research progress, this review introduces the origin and classification of CRM. We describe their crucial roles in response to inflammation during cardiac ischemic injury, to provide new ideas for improving the clinical prognosis of myocardial infarction.
-

-
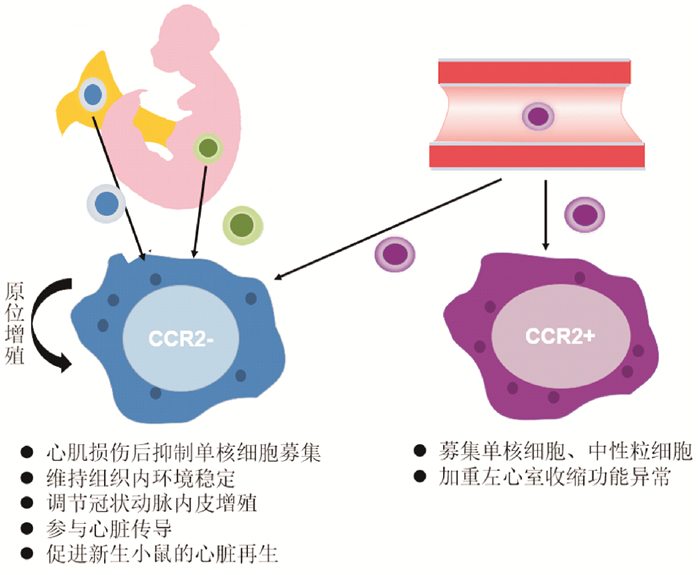
表 1 人和小鼠体内的巨噬细胞表面标志物
Table 1. Surface markers of the macrophage in human and mices
种属 表型 
CCR2- MHCⅡhi Ly6c- CX3CR1hi CD206int CD11clow(R1)
CCR2- MHCⅡlow Ly6c- CX3CR1int CD206hi CD11clow(R2)
CCR2- MHCⅡhi/low Ly6c+ CX3CR1hi CD206hi/int CD11clow(R3)
CCR2+ MHCⅡhi Ly6c- CX3CR1hi CD206int CD11chi(R1-CD11chi)
CCR2-HLA-DRhi巨噬细胞
CCR2+HLA-DRhi巨噬细胞 -
[1] Wang Z, Lu YL, Zhao WT, et al. Distinct origins and functions of cardiac orthotopic macrophages[J]. Basic Res Cardiol, 2020, 115(2): 8. doi: 10.1007/s00395-019-0769-3
[2] 杨帅涛, 廖杰, 杜以梅. 巨噬细胞在心室重塑中的作用[J]. 临床心血管病杂志, 2021, 37(4): 304-308. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB202104004.htm
[3] McNally EM. Cardiac Macrophages-Keeping the Engine Running Clean[J]. N Engl J Med, 2020, 383(25): 2474-2476. doi: 10.1056/NEJMcibr2030271
[4] Sansonetti M, Waleczek F, Jung M, et al. Resident cardiac macrophages: crucial modulators of cardiac(patho)physiology[J]. Basic Res Cardiol, 2020, 115(6): 77. doi: 10.1007/s00395-020-00836-6
[5] Litviňuková M, Talavera-López C, Maatz H, et al. Cells of the adult human heart[J]. Nature, 2020, 588(7838): 466-472. doi: 10.1038/s41586-020-2797-4
[6] Williams JW, Giannarelli C, Rahman A, et al. Macrophage Biology, Classification, and Phenotype in Cardiovascular Disease: JACC Macrophage in CVD Series(Part 1)[J]. J Am Coll Cardiol, 2018, 72(18): 2166-2180. doi: 10.1016/j.jacc.2018.08.2148
[7] Stremmel C, Schuchert R, Wagner F, et al. Author Correction: Yolk sac macrophage progenitors traffic to the embryo during defined stages of development[J]. Nat Commun, 2018, 9(1): 3699. doi: 10.1038/s41467-018-06065-9
[8] Epelman S, Lavine KJ, Beaudin AE, et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation[J]. Immunity, 2014, 40(1): 91-104. doi: 10.1016/j.immuni.2013.11.019
[9] Nahrendorf M, Swirski FK. Abandoning M1/M2 for a Network Model of Macrophage Function[J]. Circ Res, 2016, 119(3): 414-417. doi: 10.1161/CIRCRESAHA.116.309194
[10] Bajpai G, Schneider C, Wong N, et al. The human heart contains distinct macrophage subsets with divergent origins and functions[J]. Nat Med, 2018, 24(8): 1234-1245. doi: 10.1038/s41591-018-0059-x
[11] Bajpai G, Bredemeyer A, Li W, et al. Tissue Resident CCR2-and CCR2+ Cardiac Macrophages Differentially Orchestrate Monocyte Recruitment and Fate Specification Following Myocardial Injury[J]. Circ Res, 2019, 124(2): 263-278. doi: 10.1161/CIRCRESAHA.118.314028
[12] Lavine KJ, Pinto AR, Epelman S, et al. The Macrophage in Cardiac Homeostasis and Disease: JACC Macrophage in CVD Series(Part 4)[J]. J Am Coll Cardiol, 2018, 72(18): 2213-2230. doi: 10.1016/j.jacc.2018.08.2149
[13] Dick SA, Macklin JA, Nejat S, et al. Self-renewing resident cardiac macrophages limit adverse remodeling following myocardial infarction[J]. Nat Immunol, 2019, 20(1): 29-39. doi: 10.1038/s41590-018-0272-2
[14] Nicolás-Ávila JA, Lechuga-Vieco AV, Esteban-Martínez L, et al. A Network of Macrophages Supports Mitochondrial Homeostasis in the Heart[J]. Cell, 2020, 183(1): 94-109. e23. doi: 10.1016/j.cell.2020.08.031
[15] Grune J, Yamazoe M, Nahrendorf M. Electroimmunology and cardiac arrhythmia[J]. Nat Rev Cardiol, 2021, 18(8): 547-564. doi: 10.1038/s41569-021-00520-9
[16] Hulsmans M, Clauss S, Xiao L, et al. Macrophages Facilitate Electrical Conduction in the Heart[J]. Cell, 2017, 169(3): 510-522. e20. doi: 10.1016/j.cell.2017.03.050
[17] Gula G, Rumiński S, Niderla-Bielińska J, et al. Potential functions of embryonic cardiac macrophages in angiogenesis, lymphangiogenesis and extracellular matrix remodeling[J]. Histochem Cell Biol, 2021, 155(1): 117-132. doi: 10.1007/s00418-020-01934-1
[18] Gong R, Jiang Z, Zagidullin N, et al. Regulation of cardiomyocyte fate plasticity: a key strategy for cardiac regeneration[J]. Signal Transduct Target Ther, 2021, 6(1): 31. doi: 10.1038/s41392-020-00413-2
[19] Mouton AJ, DeLeon-Pennell KY, Rivera Gonzalez OJ, et al. Mapping macrophage polarization over the myocardial infarction time continuum[J]. Basic Res Cardiol, 2018, 113(4): 26. doi: 10.1007/s00395-018-0686-x
[20] Ma Y, Mouton AJ, Lindsey ML. Cardiac macrophage biology in the steady-state heart, the aging heart, and following myocardial infarction[J]. Transl Res, 2018, 191: 15-28. doi: 10.1016/j.trsl.2017.10.001
[21] Dick SA, Zaman R, Epelman S. Using High-Dimensional Approaches to Probe Monocytes and Macrophages in Cardiovascular Disease[J]. Front Immunol, 2019, 10: 2146. doi: 10.3389/fimmu.2019.02146
[22] Vagnozzi RJ, Maillet M, Sargent MA, et al. An acute immune response underlies the benefit of cardiac stem cell therapy[J]. Nature, 2020, 577(7790): 405-409. doi: 10.1038/s41586-019-1802-2
[23] DeBerge M, Yeap XY, Dehn S, et al. MerTK Cleavage on Resident Cardiac Macrophages Compromises Repair After Myocardial Ischemia Reperfusion Injury[J]. Circ Res, 2017, 121(8): 930-940. doi: 10.1161/CIRCRESAHA.117.311327
[24] Leuschner F, Nahrendorf M. Novel functions of macrophages in the heart: insights into electrical conduction, stress, and diastolic dysfunction[J]. Eur Heart J, 2020, 41(9): 989-994. doi: 10.1093/eurheartj/ehz159
[25] Rhee AJ, Lavine KJ. New Approaches to Target Inflammation in Heart Failure: Harnessing Insights from Studies of Immune Cell Diversity[J]. Annu Rev Physiol, 2020, 82: 1-20. doi: 10.1146/annurev-physiol-021119-034412
[26] Su Y, Gao J, Kaur P, et al. Neutrophils and Macrophages as Targets for Development of Nanotherapeutics in Inflammatory Diseases[J]. Pharmaceutics, 2020, 12(12): 1222. doi: 10.3390/pharmaceutics12121222
[27] Duncan SE, Gao S, Sarhene M, et al. Macrophage Activities in Myocardial Infarction and Heart Failure[J]. Cardiol Res Pract, 2020, 2020: 4375127.
[28] Pan Q, Xu J, Wen CJ, et al. Nanoparticles: Promising Tools for the Treatment and Prevention of Myocardial Infarction[J]. Int J Nanomedicine, 2021, 16: 6719-6747. doi: 10.2147/IJN.S328723
[29] Majmudar MD, Keliher EJ, Heidt T, et al. Monocyte-directed RNAi targeting CCR2 improves infarct healing in atherosclerosis-prone mice[J]. Circulation, 2013, 127(20): 2038-2046. doi: 10.1161/CIRCULATIONAHA.112.000116
[30] Bejerano T, Etzion S, Elyagon S, et al. Nanoparticle Delivery of miRNA-21 Mimic to Cardiac Macrophages Improves Myocardial Remodeling after Myocardial Infarction[J]. Nano Lett, 2018, 18(9): 5885-5891. doi: 10.1021/acs.nanolett.8b02578
[31] Protti A, Mongue-Din H, Mylonas KJ, et al. Bone marrow transplantation modulates tissue macrophage phenotype and enhances cardiac recovery after subsequent acute myocardial infarction[J]. J Mol Cell Cardiol, 2016, 90: 120-128. doi: 10.1016/j.yjmcc.2015.12.007
[32] Sun K, Li YY, Jin J. A double-edged sword of immuno-microenvironment in cardiac homeostasis and injury repair[J]. Signal Transduct Target Ther, 2021, 6(1): 79. doi: 10.1038/s41392-020-00455-6
-




 下载:
下载: