The predictive value of serum soluble ST2 on new atrial fibrillation after PCI in patients with STEMI
-
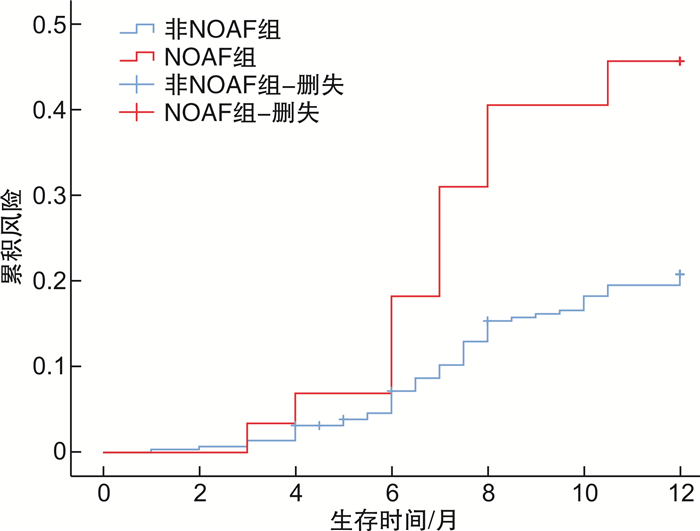
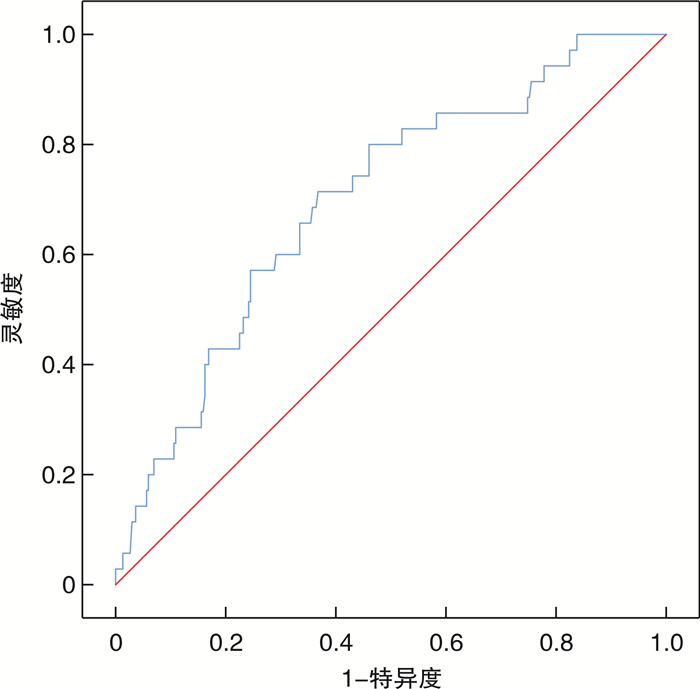
摘要: 目的 探究术前可溶性生长刺激表达基因2(soluble growth stimulation expressed gene 2,sST2)蛋白对急性ST段抬高型心肌梗死(ST-segment elevation myocardial infarction,STEMI)患者经皮冠状动脉介入治疗(percutaneous coronary intervention,PCI)术后新发心房颤动(new onset atrial fibrillation,NOAF)的预测价值。方法 连续纳入2021年1月—2021年10月就诊于徐州医科大学附属医院心内科,并于发病12 h内行直接PCI术治疗的STEMI患者,根据术后患者住院期间多次床边心电图及心电监护结果将患者分为NOAF组(35例)和非NOAF组(302例)。回顾性分析2组STEMI患者的一般临床基线资料、介入治疗相关资料,并采用二元多因素logistic回归分析筛选PCI术后NOAF的独立危险因素,采用受试者工作特征(ROC)曲线评价入院sST2水平预测PCI术后NOAF的价值,并对所有正常出院的患者进行为期1年的随访,采用Kaplan-Meier方法,绘制风险曲线,进行log-rank检验,比较NOAF组与非NOAF组患者出院后主要不良心血管事件(major adverse cardiovascular event,MACE)的发生情况。结果 2组患者在入院心室率、中性粒细胞计数、血小板计数、入院sST2、hs-CRP、VLDL-C、左室射血分数、左房内径、术后血流TIMI分级方面差异有统计学意义(P < 0.05)。二元多因素logistic回归分析显示入院sST2升高、入院心室率加快、血小板计数升高、左室射血分数降低、术后复流情况较差是导致PCI术后NOAF的独立危险因素。绘制入院sST2预测STEMI患者急诊PCI术后NOAF的ROC曲线,ROC曲线下面积为0.698(95%CI:0.610~0.786,P < 0.001),通过约登指数计算最佳截断值为46.11 ng/mL,当sST2为46.11 ng/mL时,灵敏度71.4%,特异度63.2%。并绘制风险曲线,NOAF组MACE发生率显著高于非NOAF组,行log-rank检验,P=0.014。结论 sST2是预测STEMI患者PCI术后发生NOAF的独立危险因子,当sST2≥46.11 ng/mL时,患者出现NOAF的概率较大,且NOAF组患者出院后MACE发生率高于非NOAF组。
-
关键词:
- 可溶性生长刺激表达基因2 /
- 心房颤动 /
- ST段抬高型心肌梗死 /
- 经皮冠状动脉介入治疗
Abstract: Objective To explore the predictive value of the preoperative soluble growth stimulation expressed gene 2 (sST2) protein in predicting new-onset atrial fibrillation (NOAF) after percutaneous coronary intervention (PCI) in patients with ST-segment elevation myocardial infarction (STEMI).Methods STEMI patients who were admitted to the Cardiology Department of the Affiliated Hospital of Xuzhou Medical University from January 2021 to October 2021 and received primary percutaneous coronary intervention within12 hours of the onset were divided into NOAF group (n=35) and non-NOAF group (n=302) according to the results of bedside ECG and ECG monitoring during hospitalization. The general clinical baseline data and interventional therapy-related data of the two groups of STEMI patients were retrospectively analyzed, and binary multivariate logistic regression was used to screen the independent risk factors of NOAF after PCI. The value of predicting NOAF after PCI was evaluated by the receiver operating characteristic (ROC) curve. All normallydischarged patients were followed for one year, and the risk curve was drawn by the Kaplan-Meier method. Log-rank test was performed to compare the incidence of major adverse cardiovascular events (MACE)after discharge between the NOAF group and the non-NOAF group.Results There were significant differences in heart rate, neutrophil count, platelet count, sST2, hs-CRP, VLDL-C, left ventricular ejection fraction, left atrial diameter, and postoperative blood flow TIMI grade between the two groups. Multivariate logistic regression analysis showed that increased sST2, increased heart rate, increased platelet count, decreased left ventricular ejection fraction and poor postoperative reflow were independent risk factors for new-onset atrial fibrillation after PCI. The ROC curve of admission sST2 was drawn to predict NOAF after PPCI in STEMI patients. The area under the ROC curve is 0.698(95%CI: 0.610-0.786, P < 0.001). The best cut-off value calculated by Youden's index is 46.11 ng/mL. When the sST2 is 46.11ng/mL, the sensitivity is 71.4% and the specificity is 63.2%. The risk curve was also drawn. The incidence of MACE in the NOAF groupwas significantly higher than that in the non-NOAF group. Log-rank test was performed(P=0.014).Conclusion Serum soluble sST2 is an independent risk factor for predicting the occurrence of NOAF after PCI in STEMI patients. When sST2≥46.11 ng/mL, STEMI patients after PCI are more likely to get NOAF, and the incidence of MACE after discharge in the NOAF group is higher than that in the non-NOAF group. -

-
表 1 2组患者临床资料
Table 1. clinical data of two groups of patients
例(%), X±S, M(P25, P75) 项目 非NOAF组(302例) NOAF组(35例) t/χ2值 P值 年龄/岁 63.7±12.5 66.3±9.5 1.170 0.155 男性 230(76.2) 23(65.7) 0.214 0.127 高血压 134(44.4) 17(48.6) 0.224 0.720 糖尿病 70(23.2) 6(17.1) 0.654 0.524 高脂血症 13(4.3) 3(8.6) 1.263 0.224 既往心肌梗死 36(11.9) 6(17.1) 0.784 0.415 既往PCI史 22(7.3) 4(11.4) 0.756 0.329 既往CABG史 12(4.0) 2(5.7) 0.239 0.646 吸烟史 142(47.0) 11(31.4) 3.076 0.106 左房内径/mm 34.1±4.6 35.71±5.1 1.979 <0.05 左室射血分数/% 52.1±6.6 47.5±7.3 3.888 <0.01 AST/(U·L-1) 54(43, 69) 56(37,221) 1.169 0.243 Killip分级≥Ⅱ级 39(12.9) 7(20.0) 1.336 0.294 院内MACE 脑卒中 9(3.0) 4(11.4) 6.036 <0.05 心衰 30(9.9) 8(22.9) 5.236 <0.05 心血管死亡 11(3.6) 5(14.3) 7.857 <0.05 心源性休克 44(14.6) 12(34.3) 8.799 <0.05 再次心肌梗死 9(3.0) 6(17.1) 14.793 <0.05 非计划再次血运重建 9(3.0) 6(17.1) 14.793 <0.05 入院心室率/(次·min-1) 77.9±13.4 85.6±14.0 3.209 <0.01 收缩压/mmHg 129.4±20.4 127.3±16.0 0.592 0.554 舒张压/mmHg 82.5±15.3 79.3±10.7 1.203 0.229 随机血糖/(mmol·L-1) 5.7(5.1,7.7) 5.4(5.3,6.7) 1.556 0.120 肌酐/(μmol·L-1) 63.0(53.0,74.0) 74.0(48.0,78.0) 1.797 0.072 中性粒细胞计数/(×109·L-1) 8.4±3.2 10.4±3.8 3.593 <0.01 血小板计数/(×109·L-1) 215(181, 250) 254(202, 290) 3.320 <0.01 hs-CRP/(mg·L-1) 3.5(1.1,10.4) 10.6(1.0,22.1) 2.277 <0.05 HDL-C/(mmol·L-1) 1.0±0.2 0.9±0.2 0.884 0.377 LDL-C/(mmol·L-1) 2.7±1.0 2.8±0.6 0.921 0.358 VLDL-C/(mmol·L-1) 0.5(0.4,0.6) 0.7(0.5,1.0) 2.436 <0.05 Hs-TnT峰值/(ng·L-1) 2551(945,5847) 4193(474,8496) 0.740 0.459 NT-proBNP/(pg·L-1) 955(406,1943) 1729(165,3017) 1.590 0.112 门球时间/h 6.0±3.1 6.6±3.2 1.135 0.257 sST2 /(ng·mL-1) 41.3(30.8,51.0) 54.6(42.6,62.5) 3.829 <0.01 脂蛋白a/(mg·L-1) 249(166,399) 291(203,383) 1.036 0.300 院内用药 ACEI/ARB 232(76.8) 26(74.3) 0.112 0.833 β受体阻断剂 206(68.2) 23(65.7) 0.090 0.849 利尿剂/保钾利尿剂 124(41.1) 45(42.9) 0.042 0.858 表 2 2组患者介入资料
Table 2. Interventional data of two groups of patients
例(%), M(P25, P75) 项目 非NOAF组
(302例)NOAF组
(35例)t/χ2值 P值 项目 非NOAF组
(302例)NOAF组
(35例)t/χ2值 P值 罪犯血管 0.373 0.946 病变血管数目 1.223 0.543 LM 10(3.3) 1(2.9) 1支 155(51.3) 15(42.9) LAD 141(46.7) 18(51.4) 2支 90(29.8) 11(31.4) LCX 53(17.5) 5(14.3) 3支 57(18.9) 9(25.7) RCA 98(32.5) 11(31.4) 再灌注方式 5.570 0.062 术前病变血管TIMI分级 3.463 0.073 球囊扩张成形术 36(11.9) 4(11.4) 0~1级 214(70.9) 30(85.7) 药物支架置入术 225(74.5) 21(60.0) 2~3级 88(29.1) 5(14.3) 药物球囊扩张术 41(13.6) 10(28.6) 术后血流TIMI分级 16.607 < 0.05 术中IABP 17(5.6) 3(8.6) 0.486 0.449 0~2级 18(6.0) 9(25.7) 术中血栓抽吸术 21(7.0) 3(8.6) 0.124 0.726 3级 284(94.0) 26(74.3) Gensini评分 81(47,96) 77(37,114) 0.572 0.567 表 3 以是否发生NOAF为因变量行多因素logistic回归分析
Table 3. Multivariate logistic regression analysis
因素 OR值 95%CI P值 sST2 1.030 1.008~1.051 < 0.05 入院心室率 1.037 1.006~1.069 < 0.05 中性粒细胞计数 1.101 0.978~1.240 0.110 hs-CRP 0.991 0.969~1.013 0.414 血小板计数 1.011 1.003~1.019 < 0.05 左房内径 1.044 0.950~1.147 0.370 左室射血分数 0.909 0.849~0.973 < 0.05 VLDL-C 0.527 0.155~1.793 0.305 术后血流TIMI分级 0.173 0.059~0.506 < 0.05 表 4 2组患者出院后1年MACE比较
Table 4. MACE after dischage of two groups of patient
例(%) 项目 非NOAF组
(291例)NOAF组
(30例)P值 脑卒中 6(2.1) 6(20.0) < 0.01 心衰 36(12.4) 9(30.0) < 0.05 心血管死亡 9(3.1) 4(13.3) < 0.05 心源性休克 5(1.7) 4(13.3) < 0.05 再次心肌梗死 11(3.8) 5(16.7) < 0.05 非计划再次血运重建 10(3.4) 4(13.3) < 0.05 -
[1] Ling Y, Fu C, Fan Q, et al. Triglyceride-Glucose Index and New-Onset Atrial Fibrillation in ST-Segment Elevation Myocardial Infarction Patients After Percutaneous Coronary Intervention[J]. Front Cardiovasc Med, 2022, 9: 838761. doi: 10.3389/fcvm.2022.838761
[2] Mazzone A, Scalese M, Paradossi U, et al. Development and validation of a risk stratification score for new-onset atrial fibrillation in STEMI patients undergoing primary percutaneous coronary intervention[J]. Int J Clin Pract, 2018, 72(4): e13087. doi: 10.1111/ijcp.13087
[3] Rene AG, Généreux P, Ezekowitz M, et al. Impact of atrial fibrillation in patients with ST-elevation myocardial infarction treated with percutaneous coronary intervention(from the HORIZONS-AMI[Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction]trial)[J]. Am J Cardiol, 2014, 113(2): 236-242. doi: 10.1016/j.amjcard.2013.09.016
[4] 齐晓瑜, 李敬, 张羽, 等. 可溶性生长刺激表达基因2蛋白和MHR及NLR在重症心力衰竭中的相关性研究[J]. 临床急诊杂志, 2022, 23(4): 283-288. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZLC202204013.htm
[5] Chirinos JA, Orlenko A, Zhao L, et al. Multiple Plasma Biomarkers for Risk Stratification in Patients With Heart Failure and Preserved Ejection Fraction[J]. J Am Coll Cardiol, 2020, 75(11): 1281-1295.
[6] Saez-Maleta R, Merino-Merino A, Gundin-Menendez S, et al. sST2 and Galectin-3 genotyping in patients with persistent atrial fibrillation[J]. Mol Biol Rep, 2021, 48(2): 1601-1606. doi: 10.1007/s11033-021-06150-1
[7] 中华医学会心血管病学分会, 中华心血管病杂志编辑委员会. 急性ST段抬高型心肌梗死诊断和治疗指南(2019)[J]. 中华心血管病杂志, 2019, 47(10): 766-783. https://www.cnki.com.cn/Article/CJFDTOTAL-GWXX201504001.htm
[8] Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS[J]. Eur Heart J, 2016, 37(38): 2893-2962. doi: 10.1093/eurheartj/ehw210
[9] Cozac DA, Lakatos EK, Demjen Z, et al. The CHA2DS2-VASc Score Predicts New-Onset Atrial Fibrillation and Hemodynamic Complications in Patients with ST-Segment Elevation Myocardial Infarction Treated by Primary Percutaneous Coronary Intervention[J]. Diagnostics(Basel), 2022, 12(10): 2396.
[10] Burstein B, Nattel S. Atrial fibrosis: mechanisms and clinical relevance in atrial fibrillation[J]. J Am Coll Cardiol, 2008, 51(8): 802-809. doi: 10.1016/j.jacc.2007.09.064
[11] Boralkar KA, Kobayashi Y, Amsallem M, et al. Value of Neutrophil to Lymphocyte Ratio and Its Trajectory in Patients Hospitalized With Acute Heart Failure and Preserved Ejection Fraction[J]. Am J Cardiol, 2020, 125(2): 229-235. doi: 10.1016/j.amjcard.2019.10.020
[12] 赵丹莉, 王宪沛, 张智文, 等. 频发房性期前收缩与老年急性ST段抬高型心肌梗死患者新发心房颤动的关系及其预测价值研究[J]. 临床心血管病杂志, 2021, 37(1): 57-61. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB202101013.htm
[13] 毕昌龙, 张侃迪, 周恩, 等. 急性心肌梗死患者住院期间新发心房颤动危险因素分析及预测模型的建立[J]. 临床心血管病杂志, 2021, 37(8): 731-735. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB202108011.htm
[14] 陈东, 董玲玲, 黄宏, 等. 静息心率与射血分数降低性心力衰竭伴心房颤动患者预后的关联[J]. 中华高血压杂志, 2021, 29(10): 986-990. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGGZ202110022.htm
[15] Larmann J, Handke J, Scholz AS, et al. Preoperative neutrophil to lymphocyte ratio and platelet to lymphocyte ratio are associated with major adverse cardiovascular and cerebrovascular events in coronary heart disease patients undergoing non-cardiac surgery[J]. BMC Cardiovasc Disord, 2020, 20(1): 230.
[16] Rabkin SW, Tang JKK. The utility of growth differentiation factor-15, galectin-3, and sST2 as biomarkers for the diagnosis of heart failure with preserved ejection fraction and compared to heart failure with reduced ejection fraction: a systematic review[J]. Heart Fail Rev, 2021, 26(4): 799-812. https://www.sciencedirect.com/science/article/pii/S0009898115001771
[17] Aimo A, Januzzi JJ, Bayes-Genis A, et al. Clinical and prognostic significance of sST2 in heart failure: JACC review topic of the week[J]. J Am Coll Cardiol, 2019, 74(17): 2193-2203. https://www.sciencedirect.com/science/article/abs/pii/S1071916413011731
[18] Barnett O, Halkevych M, Labinska O, et al. The role of NT-probnp and ST2 biomarkers in patients with acute coronary syndrome[J]. Wiad Lek, 2022, 75(1): 34-38.
[19] Tan R, Yu H, Han X, et al. Circulating Soluble Suppression of Tumorigenicity 2 Predicts Recurrence After Radiofrequency Ablation of Persistent Atrial Fibrillation[J]. Front Cardiovasc Med, 2021, 8: 653312.
[20] 杨敏, 肖模超. 心房颤动患者左心房形态结构及功能与血栓形成关系的研究进展[J]. 临床心血管病杂志, 2023, 39(2): 103-107. https://lcxxg.whuhzzs.com/article/doi/10.13201/j.issn.1001-1439.2023.02.006
[21] Budzianowski J, Hiczkiewicz J, Burchardt P, et al. Predictors of atrial fibrillation early recurrence following cryoballoon ablation of pulmonary veins using statistical assessment and machine learning algorithms[J]. Heart Vessels, 2019, 34(2): 352-359.
[22] Jortveit J, Pripp AH, Langørgen J, et al. Poor adherence to guideline recommendations among patients with atrial fibrillation and acute myocardial infarction[J]. Eur J Prev Cardiol, 2019, 26(13): 1373-1382.
[23] Chiang CH, Hung WT, Huang WC, et al. The risk of stroke after acute myocardial infarction in patients with and without atrial fibrillation: A nationwide cohort study[J]. J Chin Med Assoc, 2021, 84(12): 1126-1134.
[24] Khalfallah M, Elsheikh A. Incidence, predictors, and out-comes of new-onset atrial fibrillation in patients with ST-elevation myocardial infarction[J]. Ann Noninvasive Electrocardiol, 2020, 25(4): e12746.
-




 下载:
下载: