Association between high-sensitivity Glasgow prognostic score and long-term death for acute heart failure with preserved ejection fraction
-
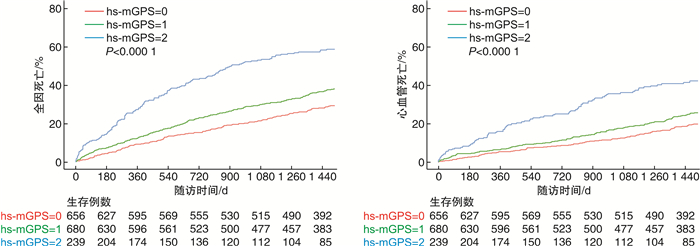
摘要: 目的 探索高敏感格拉斯哥评分(high-sensitivity modified Glasgow prognostic score,hs-mGPS)与急性射血分数保留型心力衰竭(heart failure with preserved ejection fraction,HFpEF)住院患者长期预后的关联性。方法 从重大慢病国家注册登记研究心力衰竭前瞻队列中选取HFpEF住院患者,纳入有基线白蛋白和高敏C反应蛋白(high-sensitivity C-reactive protein,hs-CRP)检测结果的患者,计算其hs-mGPS评分。依据hs-mGPS评分,将所纳入患者分为3组:0分组(hs-CRP≤3 mg/L)、1分组(hs-CRP>3 mg/L且白蛋白≥35 g/L)、2分组(hs-CRP>3 mg/L且白蛋白 < 35 g/L),探索HFpEF患者的hs-mGPS评分与长期全因死亡及心血管死亡的关联性。结果 共纳入1 575例急性HFpEF患者,中位年龄71(63,79)岁,女性占比50.7%;0分组656例(41.6%),1分组680例(43.2%),2分组239例(15.2%)。出院后患者的中位随访时间为4.54(4.09,4.39)年,642例(40.8%)患者发生全因死亡,其中400例(25.4%)发生心血管死亡。多因素Cox分析显示,1分组与2分组患者发生全因死亡风险分别是0分组患者的1.28倍(HR=1.28,95%CI:1.07~1.53,P < 0.01)和2.21倍(HR=2.21,95%CI:1.77~2.76,P < 0.01);此外,2分组患者发生心血管死亡风险是0分组患者的2.32倍(HR=2.32,95%CI:1.75~3.07,P < 0.01)。结论 hs-mGPS与急性HFpEF住院患者的长期预后密切相关,其评分越高,出院后全因死亡与心血管死亡风险也越高。
-
关键词:
- 射血分数保留型心力衰竭 /
- 高敏感格拉斯哥评分 /
- 死亡 /
- 长期预后
Abstract: Objective To analyze the relationship between high-sensitivity modified Glasgow prognostic score(hs-mGPS) and long-term death after discharge for heart failure with preserved ejection fraction(HFpEF).Methods Data were obtained from the patients hospitalized for acute HFpEF in the China Patient-centered Evaluative Assessment of Cardiac Events(PEACE) -Prospective Heart Failure Study. Patients with high-sensitivity C-reactive protein(hs-CRP) and albumin data were enrolled in this analysis. Patients were divided into hs-mGPS=0 group(hs-CRP ≤ 3 mg/L), hs-mGPS=1 group(hs-CRP>3 mg/L and albumin ≥ 35 g/L), hs-mGPS=2 group(hs-CRP>3 mg/L and albumin < 35 g/L). We analyzed the association between hs-mGPS and risks of long-term all-cause death and cardiovascular death in patients with HFpEF.Results The study included 1 575 acute HFpEF patients; the median age was 71(63, 79) years, and 50.7% were women. There were 656(41.6%) patients in the hs-mGPS=0 group, 680(43.2%) in the hs-mGPS=1 group, and 239(15.2%) in the hs-mGPS=2 group. The median follow-up time was 4.54(4.09, 4.39) years. A total of 642(40.8%) patients died, among whom 400(24.4%) died for cardiovascular reasons. In multivariable Cox analyses, the risk of all-cause death in the hs-mGPS=1 and hs-mGPS=2 group were 1.28 times(HR=1.28, 95%CI: 1.07-1.53, P < 0.01) and 2.21 times(HR=2.21, 95%CI: 1.77-2.76, P < 0.01) than that in the hs-mGPS=0 group. Besides, the risk of cardiovascular death in the hs-mGPS=2 group was 2.32 times(HR=2.32, 95%CI: 1.75-3.07, P < 0.01) than that in the hs-mGPS=2 group.Conclusion hs-mGPS is associated with prognosis in patients with acute HFpEF, and increasing hs-mGPS indicates higher risks of all-cause death and cardiovascular death. -

-
表 1 患者基本特征
Table 1. Basic characteristics
例(%), M(P25, P75) 项目 全体(1 575例) 0分组(656例) 1分组(680例) 2分组(239例) P值 年龄/岁 71(63,79) 71(62,78) 72(64,78) 71(63,80) 0.12 女性 799(50.7) 333(50.8) 350(51.5) 116(48.5) 0.74 收缩压/mmHg 137(120,153) 136(120,151) 138(121,153) 138(118,157) 0.42 舒张压/mmHg 80(70,90) 80(70,90) 80(70,90) 78(68,90) 0.03 心率/(次/min) 83(70,98) 80(68,94) 86(74,100) 87(76,101) < 0.01 NYHA分级 < 0.01 Ⅱ 342(21.7) 191(29.2) 131(19.3) 20(8.4) Ⅲ 763(48.4) 320(48.8) 324(47.7) 119(49.8) Ⅳ 470(29.8) 145(22.1) 225(33.1) 100(41.8) 病史 冠心病 920(58.4) 389(59.3) 411(60.4) 120(50.2) 0.02 高血压 1058(67.2) 437(66.6) 467(68.7) 154(64.4) 0.45 心房颤动 707(44.9) 290(44.2) 327(48.1) 90(37.7) 0.02 肾功能下降 468(29.7) 183(27.9) 196(28.8) 89(37.2) 0.02 心肌梗死 253(16.1) 93(14.2) 122(17.9) 38(15.9) 0.17 入院时合并肺炎 328(20.8) 77(11.7) 164(24.1) 87(36.4) < 0.01 慢性阻塞性肺疾病 385(24.4) 125(19.1) 192(28.2) 68(28.5) < 0.01 脑卒中 425(27.0) 176(26.8) 193(28.4) 56(23.4) 0.33 心脏瓣膜病 339(21.5) 140(21.3) 149(21.9) 50(20.9) 0.94 贫血 359(22.8) 129(19.7) 135(19.9) 95(39.7) < 0.01 糖尿病 221(14.0) 78(11.9) 109(16.0) 34(14.2) 0.09 心衰类型 新发 559(35.5) 221(33.7) 247(36.3) 91(38.1) 0.40 慢性急性加重 1016(64.5) 435(66.3) 433(63.7) 148(61.9) 0.40 LVEF/% 60(55,65) 60(55,65) 59(55,64) 60(55,66) 0.06 NT-proBNP/(ng/L) 902(338,2 074) 637(194,1 414) 1 000(401,2 175) 2 069(742,4 650) < 0.01 出院带药 ACEI/ARB 689(43.8) 305(46.5) 300(44.1) 84(35.2) < 0.01 β受体阻滞剂 783(49.7) 358(54.6) 337(49.6) 88(36.8) < 0.01 醛固酮受体拮抗剂 801(50.9) 322(49.1) 352(51.8) 127(53.1) 0.46 1 mmHg=0.133 kPa。肾功能下降:估算肾小球滤过率 < 60 mL/min/1.73 m2;ACEI/ARB:血管紧张素转化酶抑制剂/血管紧张素Ⅱ受体拮抗剂。 表 2 各组患者长期死亡率比较
Table 2. Comparison of long-term mortality rates among groups
例(%) 项目 全体(1 575例) 0分组(656例) 1分组(680例) 2分组(239例) P值 全因死亡 642(40.8) 211(32.1) 283(41.6) 148(61.9) < 0.01 心血管死亡 400(25.4) 136(20.7) 172(25.3) 92(38.5) < 0.01 表 3 hs-mGPS评分与长期死亡的Cox回归分析
Table 3. Cox regression analysis of hs-mGPS score and long-term mortality
HR(95%CI) 类型 0分组
(656例)1分组
(680例)2分组
(239例)全因死亡 单因素模型 1 1.38(1.15~1.64) 2.67(2.16~3.29) 多因素模型1 1 1.32(1.11~1.58) 2.68(2.17~3.31) 多因素模型2 1 1.28(1.07~1.53) 2.21(1.77~2.76) 心血管死亡 单因素模型 1 1.30(1.04~1.63) 2.65(2.03~3.45) 多因素模型1 1 1.35(1.00~1.57) 2.68(2.05~3.50) 多因素模型2 1 1.20(0.96~1.51) 2.32(1.75~3.07) 模型1调整因素:性别、年龄;模型2调整因素:性别、年龄、收缩压、高血压、冠心病、心房颤动、心脏瓣膜病、心衰类型、入院时合并肺炎、贫血、NT-proBNP、出院带药。 表 4 不同年龄HFpEF患者hs-mGPS评分与长期死亡风险的关联差异
Table 4. The association between hs-mGPS scores and long-term mortality risk in HFpEF patients with different ages
HR(95%CI) 项目 1分组vs 0分组 2分组vs 0分组 交互作用P值 全因死亡 年龄 0.396 < 70岁 1.51(1.08~2.11) 2.67(1.82~3.91) ≥70岁 1.20(0.97~1.48) 2.02(1.54~2.64) 心血管死亡 年龄 0.591 < 70岁 1.38(0.90~2.12) 2.87(1.76~4.68) ≥70岁 1.15(0.88~1.51) 2.13(1.52~2.98) -
[1] Hao G, Wang X, Chen Z, et al. Prevalence of heart failure and left ventricular dysfunction in China: the China Hypertension Survey, 2012-2015[J]. Eur J Heart Fail, 2019, 21(11): 1329-1337. doi: 10.1002/ejhf.1629
[2] Wang H, Chai K, Du M, et al. Prevalence and incidence of heart failure among urban patients in China: a national population-based analysis[J]. Circ Heart Fail, 2021, 14(10): e008406. doi: 10.1161/CIRCHEARTFAILURE.121.008406
[3] 张丽华, 蒲博轩, 陈腾, 等. 急性心力衰竭住院患者出院后2年内生存情况分析[J]. 中国循环杂志, 2023, 38(3): 284-290.
[4] 王华, 刘宇佳, 杨杰孚. 心力衰竭流行病学[J]. 临床心血管病杂志, 2023, 39(4): 243-247. https://lcxxg.whuhzzs.com/article/doi/10.13201/j.issn.1001-1439.2023.04.001
[5] McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure[J]. Eur Heart J, 2021, 42(36): 3599-3726. doi: 10.1093/eurheartj/ehab368
[6] Ando K, Sakamoto S, Saito S, et al. Prognostic value of High-Sensitivity Modified Glasgow Prognostic Score in castration-resistant prostate cancer patients who received docetaxel[J]. Cancers(Basel), 2021, 13(4): 773.
[7] Wu TH, Tsai YT, Chen KY, et al. Utility of High-Sensitivity Modified Glasgow Prognostic Score in cancer prognosis: a systemic review and meta-analysis[J]. Int J Mol Sci, 2023, 24(2): 1318. doi: 10.3390/ijms24021318
[8] Wu TH, Tsai YT, Chen KY, et al. Utility of High-Sensitivity Modified Glasgow Prognostic Score in cancer prognosis: a systemic review and meta-analysis[J]. Int J Mol Sci, 2023, 24(2): 1318. doi: 10.3390/ijms24021318
[9] Pellicori P, Zhang J, Cuthbert J, et al. High-sensitivity C-reactive protein in chronic heart failure: patient characteristics, phenotypes, and mode of death[J]. Cardiovasc Res, 2020, 116(1): 91-100. doi: 10.1093/cvr/cvz198
[10] 高蓉蓉, 徐芳, 祝绪, 等. 全血细胞衍生的炎症标志物对急性心力衰竭患者的长期预后价值[J]. 临床心血管病杂志, 2022, 38(12): 980-987. https://lcxxg.whuhzzs.com/article/doi/10.13201/j.issn.1001-1439.2022.12.010
[11] Briasoulis A, Androulakis E, Christophides T, et al. The role of inflammation and cell death in the pathogenesis, progression and treatment of heart failure[J]. Heart Fail Rev, 2016, 21(2): 169-176. doi: 10.1007/s10741-016-9533-z
[12] El Iskandarani M, El Kurdi B, Murtaza G, et al. Prognostic roleof albumin level in heart failure: a systematic review and meta-analysis[J]. Medicine, 2021, 100(10): e24785. doi: 10.1097/MD.0000000000024785
[13] Mene-Afejuku TO, Moisa EA, Akinlonu A, et al. The relevance of serum albumin among elderly patients with acute decompensated heart failure[J]. J Geriatr Cardiol, 2019, 16(7): 522-528.
[14] Huang RC, Lillycrop KA, Beilin LJ, et al. Epigenetic age acceleration in adolescence associates with BMI, inflammation, and risk score for middle age cardiovascular disease[J]. J Clin Endocrinol Metab, 2019, 104(7): 3012-3024. doi: 10.1210/jc.2018-02076
[15] Hu Y, Wang X, Huan J, et al. Effect of dietary inflammatory potential on the aging acceleration for cardiometabolic disease: A population-based study[J]. Front Nutr, 2022, 9: 1048448. doi: 10.3389/fnut.2022.1048448
[16] Itagaki T, Motoki H, Otagiri K, et al. Inflammation-based assessment for the risk stratification of mortality in patients with heart failure[J]. Sci Rep, 2021, 11(1): 14989. doi: 10.1038/s41598-021-94525-6
-




 下载:
下载: